
This is again the nature of QM.The Uncertainty Principle, introduced by Heisenberg in 1927, applies to observations of the properties of the quantum world, which is typically microscopic in scale. Pauli exclusion principle, the electron is a fermion, like the quarks in the nucleus, and as they would get closer and closer, you would get to a limit where they simply would not be able to get closer, and your technology would not work again. Heisenberg uncertainty principle, as you would start restricting the electron to a smaller position (QM orbital) around the nucleus, it would gain momentum, so its momentum would be known with less certainty, and you would lose information instead of gaining. 
Now, to get a more precise measurement of the electron's position you would have to want to restrict it to a smaller and smaller space around the nucleus. But we already know that with today's technology, so what would be the advancement? What you would get, even if you would get more info about the electron's position with some advanced technology, is that the electron exists around the nucleus. This method would not be able to restrict the electron to a smaller space then what is described by its wavefunction, the description of the probability distribution of the electron around the nucleus. Now since the electron exists around the nucleus, you would start shooting photons towards the nucleus, and expect the electron to start having a more and more defined position (in your theory). So in your setup the electron should exist around a nucleus. And no, you cannot have an electron at rest in an experiment, and you cannot have an electron in vacuum at rest where you shoot photons towards the electron. In this case you most probably would have to have an electron existing around a nucleus, where we would have at least some information about the position of the nucleus. Now, to do that, we at least would have to have a certain part of space as the probable location for the electron. Now you are suggesting that we should try to shoot photons a towards the electron from all directions. The electron exist around the nucleus at a certain energy level as per QM everywhere with certain probabilities. The most important thing is that the nature of our universe on the micro level is probabilities. The phenomena of the electron not being a classical billiard ball, but a fuzzy point-particle, that is based on the nature of our universe, and QM describes it the most accurately. The problem is not with out measuring apparatus (this is what your question suggests). Our models predict that (and the experiments verify):Īs per the SM, the electron is an elementary point particle, does not have any spatial etension or internal structure.Īs per the QM, the electron is in an orbital (not a classical orbit), which means that the electron is existint around the nucleus at a certain energy level as per QM. It is very important to distinct between a classical orbit (that you are maybe using here) from the QM orbital.  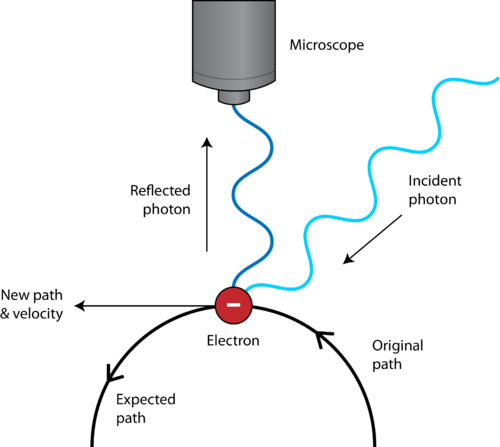
These phenomena include the orbit of the electron. Our currently accepted theory, the Standard Model, together with QM, has been validated by numerous experiments, and the theory fits the data the best, that is why we use these theories as our underlying methods to model the physical phenomena.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
